Raman spectroscopy
A current area of research into improving the speed and accuracy of the identification process of different strains of bacteria in a clinical environment is using the phenomenon of the inelastic scattering of light, Raman spectroscopy. This was first observed by Sir C.V. Raman in 1928 using sunlight, from which point the process has developed through using mercury emission as the excitation source to the modern practice of using laser radiation.
In modern Raman spectroscopy the benefits of this method with regards to the typing of bacteria are at the forefront of research into combating problems such as the formation of bacterial biofilms on implant surfaces.Staphylococcus epidermidis and S. aureus are common causes of these infections and biofilms, so it is important to be able to type these and similar strains of bacteria in order to effectively treat the infection. Where Raman spectroscopy demonstrates clear benefits over existing chemical means of identification such as pulsed-field gel electrophoresis (PFGE) is in the speed of the identification process, which is particularly important when considering the formation of biofilms, which once established can be very difficult to treat as the colonies have formed adhered to the surface under a protective matrix of polysaccharides.
It has been shown in many studies that Raman spectroscopy is capable to rapidly identify / discriminate biological samples, and medically relevant microorganisms and bacteria. In this context, recent reviews provide valuable information on Raman spectroscopy and its use to detect and identify important molecular complexes in biological samples. And, based on extensive studies, in 2007 the Raman Research Group at Gent University published the first database for Raman features of biological samples.
It has been shown that it is possible to use Raman spectroscopy for the accurate typing of bacteria, however the procedures demonstrated still have limitations with regards to the analysis of the results such that the procedure would be reproducible in a clinical environment.
Raman spectroscopy is based on the phenomenon of Raman scattering of monochromatic light, which is the inelastic scattering of a photon. When there is monochromatic light incident on a target there are several possibilities for the incoming photons, if they have sufficient energy the molecules of the target can be raised to an excited electronic state and the photons absorbed, they can pass through the target without interacting or they can undergo elastic or inelastic scattering. In the case of elastic scattering, a molecule of the sample is excited to a virtual state by absorbing the incoming photon, and then drops back down to its original state, emitting a photon of the same energy as the one that caused the excitation. This is known as Rayleigh scattering. Overall, the molecule does not absorb energy from or give energy to the scattered photon.
Elastic scattering is the type of scattering that occurs most often when light is incident upon a target. In Raman spectroscopy however, inelastic scattering is exploited despite the low frequency occurrence. In this case too the molecule is excited to a virtual energy state, however this time when it relaxes it returns to a different vibrational energy state than the one that it started from. Therefore the energy of the photon emitted during the relaxation is different to the energy of the photon that caused the excitation in the first place. The scattered photon consequently has a different frequency than the excitation source and this is what produces the Raman spectrum, a plot of the frequency shift between incident and scattered light.
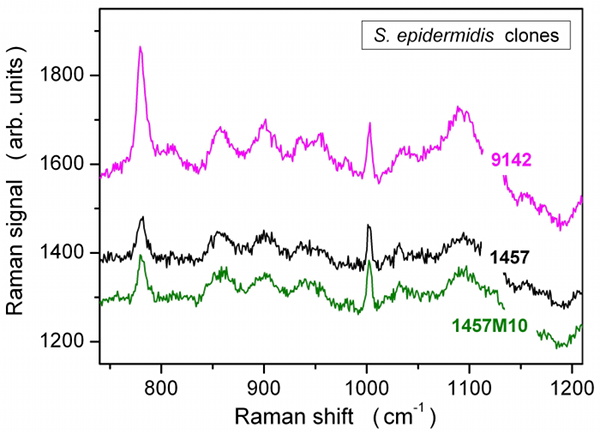
Typical Raman spectra for the three S epidermidis strains (1457, 1457M10, and 9142), cultured for 24 hours, from a single raster point within individual bacterial colonies. Traces are offset to each other for clarity.
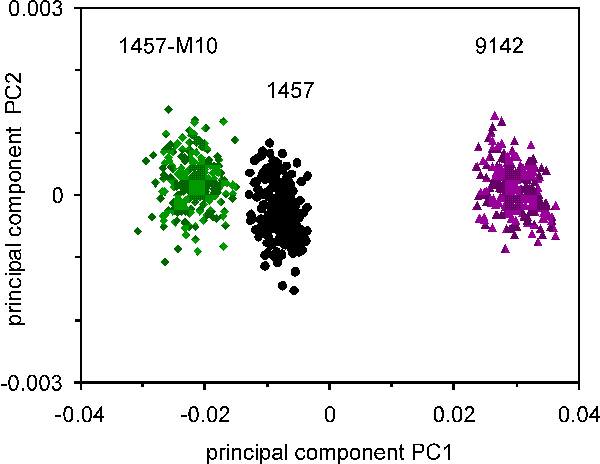
Plot of principle component relation for the three S epidermidis strains (1457, 1457M10, and 9142), cultured for 24 hours. Spectra were obtained from multiple raster points of individual bacterial colonies. The separation of the clusters of spectra associated with different natural strains is significant.
Our findings show that Raman spectroscopy might be capable in aiding rapid identification of clinically important bacterial strains, so that time and cost of patient treatment related to virulent infection could be significantly reduced. However, systematic studies are still required to investigate biofilm formation and detection further, in order to improve on the discriminatory power between biofilm-positive and biofilm-negative strains. Such studies are currently under way in our laboratories, exploiting a selection of Raman spectroscopy approaches. Specifically, we aim to be able to identify and characterise bacterial cells using techniques such as Raman tweezers.
Kontakt: Mgr.Ota Samek,Dr.
E-mail: osamek@isibrno.cz, tel: + 420 541 514 127
Reference:
Peck E., Master Degree Thesis, Swansea University, UK, 2009.
Samek O., Telle H.H., Harris L.G., Bloomfield M., Mack D.: Raman spectroscopy for rapid discrimination of Staphylococcus epidermidis clones related to medical device-associated infections. Laser Physics Letters 5 (2008), 465-470.
Samek O., Al-Marashi J.F.M., Telle H.H.: The potential of Raman spectroscopy for the identification of biofilm formation by Staphylococcus epidermidis. Laser Physics Letters 7 (2010), 378-383.
Send comments to webmaster
Last modification: 20 Apr 2010

